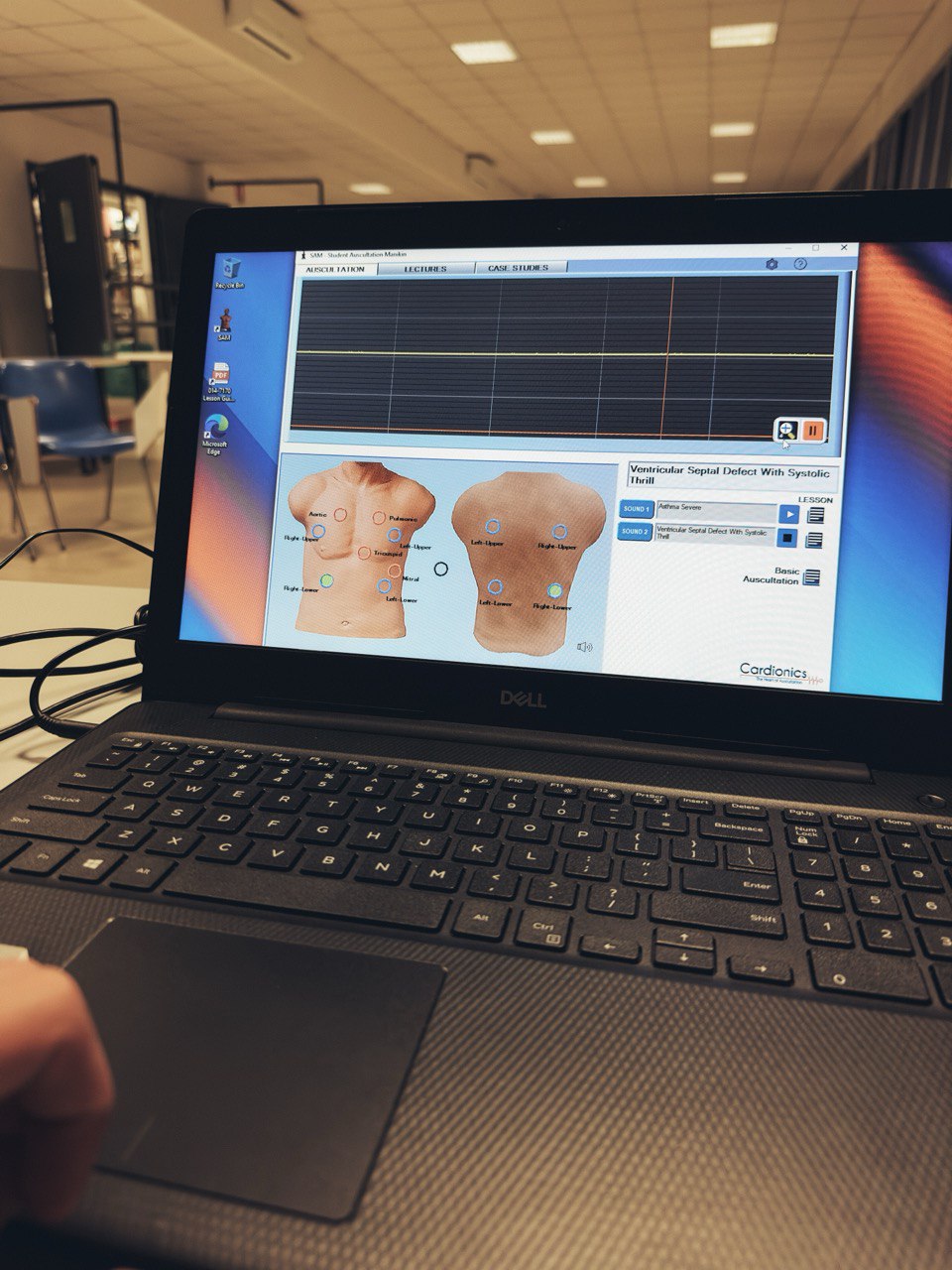
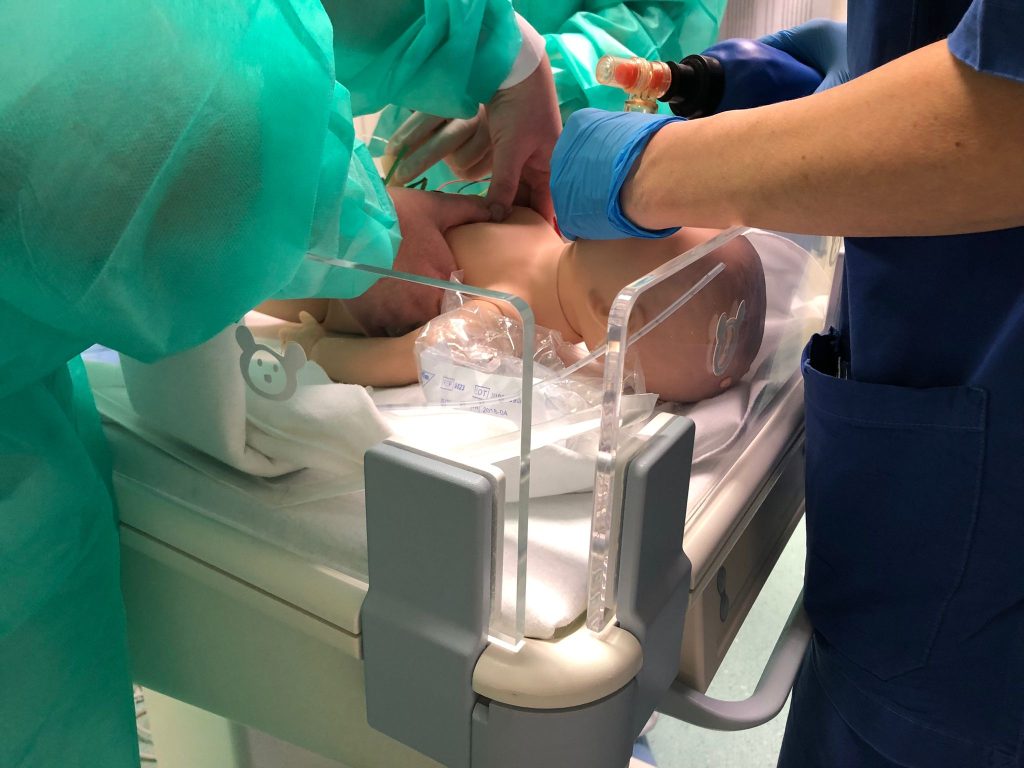
How is an ECMO simulator created? Two inventors explain it to us directly, shedding light on some processes that are often obscure to the public
There is no handbook of medical training simulator inventions, therefore as developers we have to come up with our own methods. For the developers of the Human Patient Simulator at the University of Florida that meant using the intuition and collective intelligence of a well-integrated team of clinical educators and engineers. The developers of the Lucina Childbirth simulator at the University of Porto looked at a monograph on the design of combat helicopter simulators by Farmer et al. (1999), who use four coupled simulation and simulator design phases:
Training Needs Analysis (TNA),
Training Program Design (TPD),
Training Media Specification (TMS), and
Training Evaluation (TE).
For the design of acute care training simulators, TNA requires a clinical perspective, TPD an educational perspective, and TMS an engineering perspective. TE will establish if these three perspectives were successfully combined or not. Three adaptations were made to this method:
- During TNA, a detailed analysis of critical incidents was included to justify the development of a new simulator. In the case of the childbirth simulator, these were the Confidential inquiries into stillbirths and deaths in infancy.
- Farmer et al. introduced the concept of “Norm Scenarios” only in the TE phase. For the development of the childbirth simulator, this concept was expanded to a full list of clinical conditions that trainees should be able to recognize and manage. This list was then used throughout the design process. Examples for the childbirth simulator are fetal position related blockage of flow through the umbilical cord, and post-partum hemorrhage.
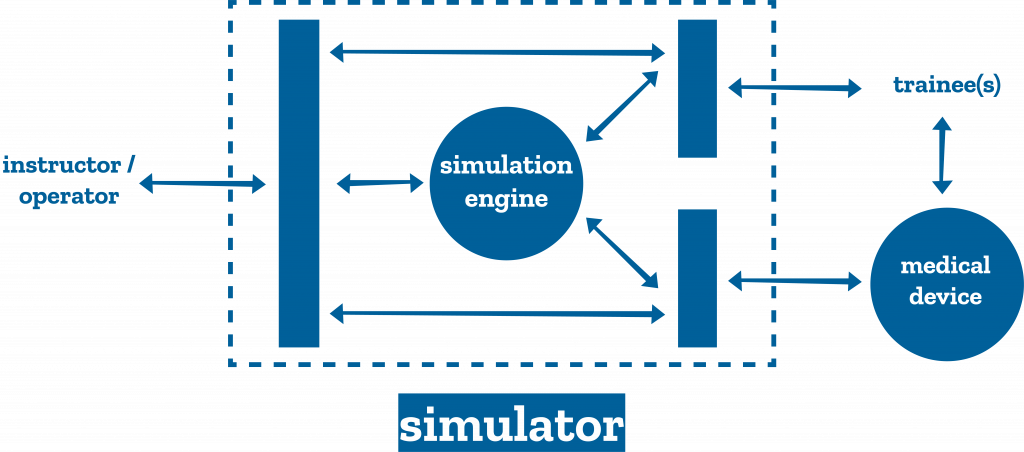
- The functional units of an acute care simulator, subject to design in the TMS phase, were identified, Figure 1.
When Timothy Antonius started work on a training simulator for neonatal Extracorporeal Membrane Oxygenation (ECMO), a technology intensive external artificial heart-lung, he had a mostly verbal and fragmented account of the above-described design method, but was able to follow its steps. Doctors and nurses are trained in treating newborns with serious respiratory problems using ECMO. Rates of serious, potentially life-threatening complications with this intervention vary between 30 and 70%. Traditional training took place on lambs. Norm scenarios were derived from the guidelines of the Extracorporeal Life Support Organization for training and continuing education. Examples include: venous side air entrainment, hypovolemic incidents through unexpected bleeding, and cavitation from a kinked cannula. The proposed one-day course starts with a short lecture on ECMO pathophysiology, followed by hands-on training.
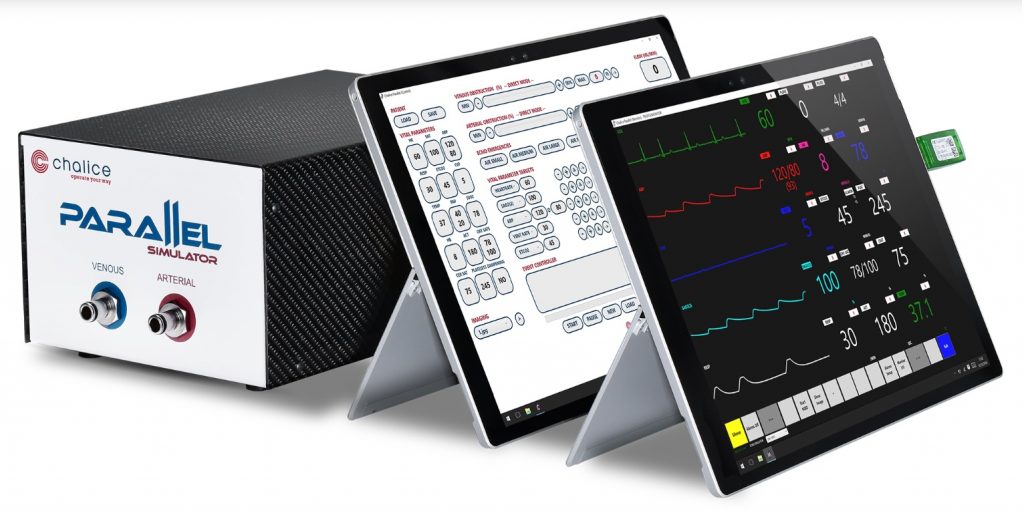
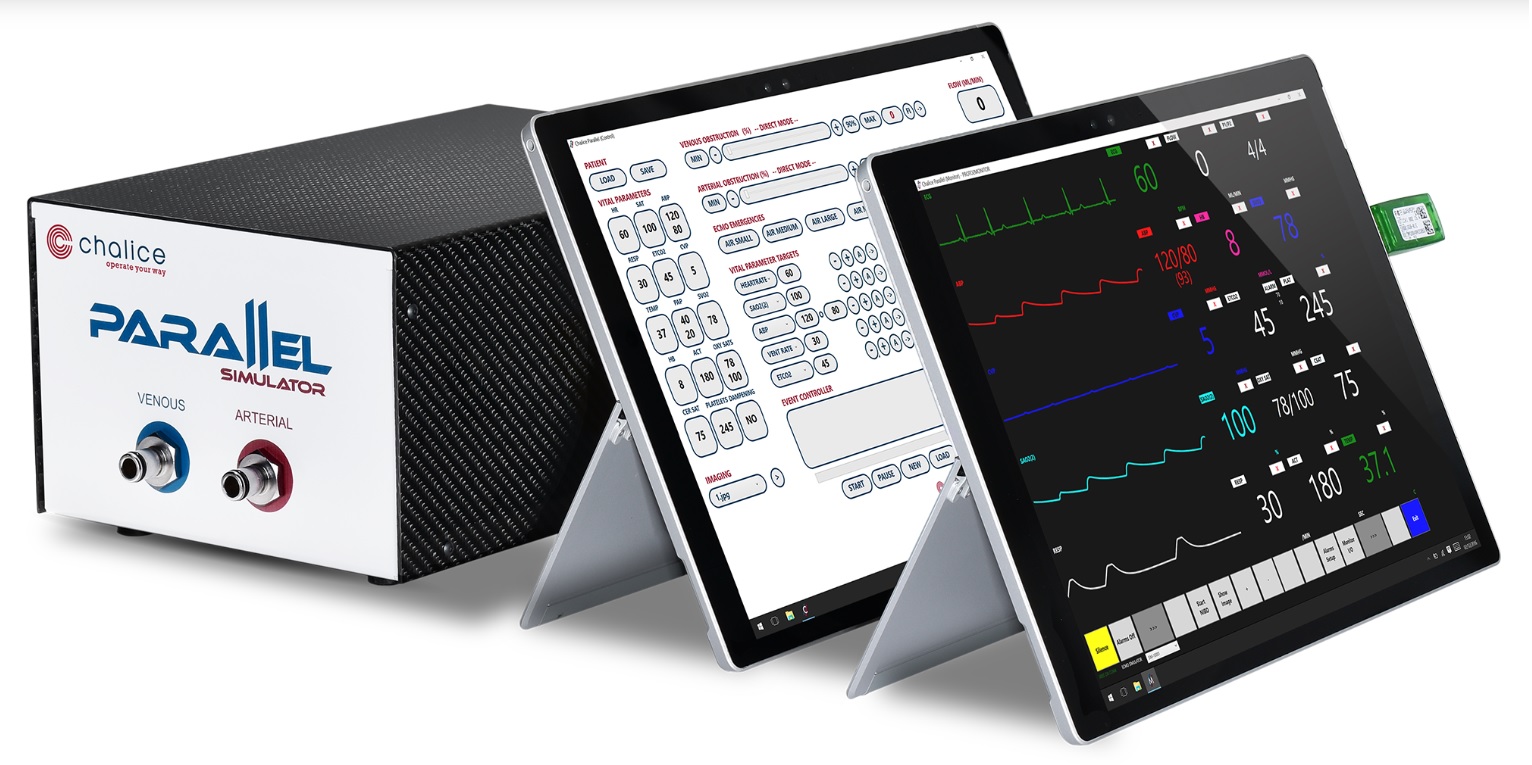
In team simulation sessions, trainees are confronted with four scenarios followed by a half-hour debriefing per scenario. The simulator and other training media are designed keeping the requirements of the training program in mind. The trainees interact with a baby mannequin for cannulation and assessment of clinical signs, and with a real ECMO machine. The simulator includes an inline system with venous and arterial connectors. Two wireless tablets contain the instructor/operator interface, and a clinical monitor emulator, respectively [Figure2].
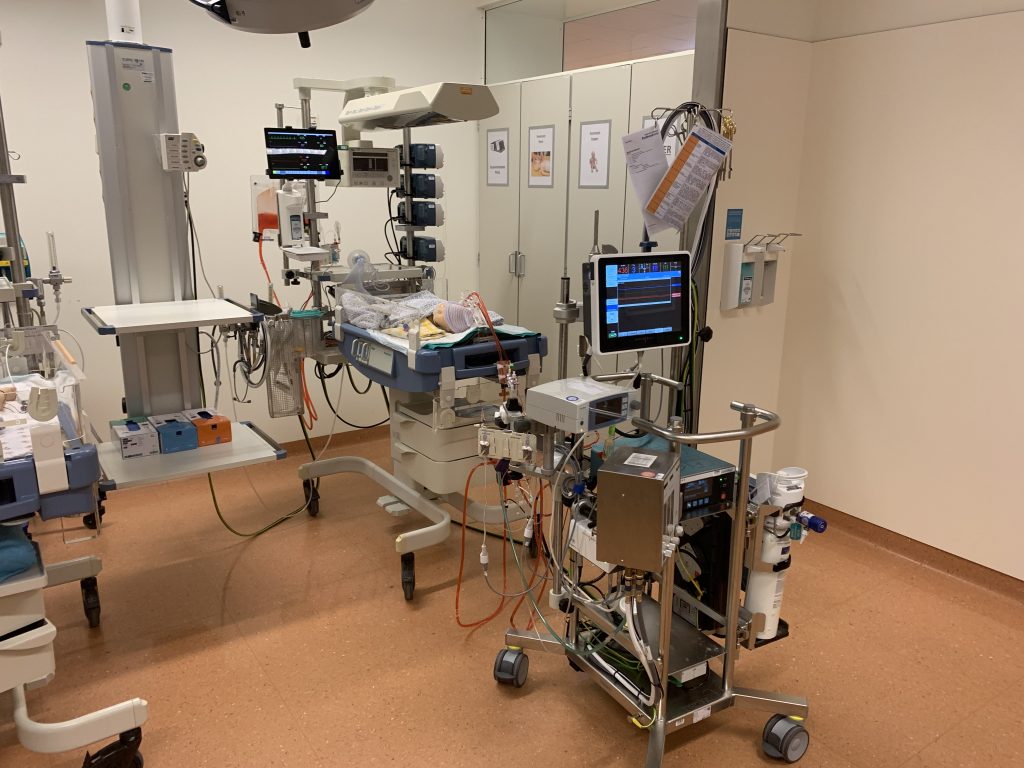
Using a monitor emulator, rather than real clinical monitors makes the system customizable and more affordable. The instructor can write, select, launch, and modulate the severity of clinical scenarios. The simulation engine consists of models of neonatal cardiorespiratory physiology, modulated by the instructor. Figure 3 shows the complete system in-situ.
The simulator is commercialized by ECMO oxygenator manufacturer Chalice Medical, Ltd, Worksop, UK. Educational impact studies are encouraged by the developer and the manufacturer. Analysis of the potential market and optimization of technology for manufacturing and maintenance also played a role in the design, but are not further discussed here.
References
Farmer E, van Rooij J, Riemersma J, Jorna P, Moraal J: Handbook of simulator-based training, Ashgate, Aldershot, 1999.
Van Meurs W: Modeling and simulation in biomedical engineering: Application to cardiorespiratory physiology, New York, McGraw-Hill, 2011.
READ ALSO